| Table of Contents |  |
|
Original Article
| ||||||
| Patient radiation dose during pelvic artery embolization used for controlling postpartum hemorrhaging | ||||||
| Izumi Torimoto1, Shigeo Takebayashi1, Zenjiro Sekikawa1, Akitoshi Nishii1, Soichiro Obata2, Shigeru Aoki2 | ||||||
|
1The Departments of Diagnostic Radiology, Yokohama City University Medical Center, Yokohama, Japan.
2The Perinatal Center for Maternity and Neonate, Yokohama City University Medical Center, Yokohama, Japan. | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article |
| Torimoto I, Takebayashi S, Sekikawa Z, Nishii A, Obata S, Aoki S. Patient radiation dose during pelvic artery embolization used for controlling postpartum hemorrhaging. Edorium J Radiol 2016;2:20–26. |
|
Abstract
|
|
Aims:
To evaluate the radiation exposure in pelvic artery embolization (PAE) with N-butyl cyanoacrylate (NBCA) used for controlling postpartum hemorrhaging.
Methods: This retrospective study included 65 consecutive PAE procedures for controlling hemorrhaging in 60 postpartum patients who underwent PAE at a single institution over the last five years. The 65 PAE procedures were divided into two groups: (i) the procedure with NBCA (NBCA group) and (ii) gelatin-sponge PAE with or without coil(s) (non-NBCA group). We compared mean numbers of embolized arteries, mean fluoroscopy time, and mean dose-area product (DAP) of the two groups. Results: There were 36 (55.4%) and 29 (44.6%) procedures in the NBCA and non-NBCA groups, respectively. No significant differences in the clinical background and mean numbers of embolized arteries between the two groups were observed. The NBCA group had a shorter mean fluoroscopic time and a smaller mean DAP than the non-NBCA group (26.3±11.1 min versus 34.2±11.0 min, p = 0.011, and 43.5±19.5 Gy·cm2 versus 55.1±16.1 Gy·cm2, p = 0.003). Conclusion: Pelvic artery embolization with NBCA is superior in reducing radiation exposure in patients with postpartum hemorrhaging. | |
|
Keywords:
Cyanoacrylate, Embolization, Hemorrhage, Postpartum, Radiation exposure
| |
|
Introduction
| ||||||
|
Postpartum hemorrhaging is one of the main causes of maternal mortality; the most common causes of hemorrhaging are uterine atony and lower genital tract laceration [1]. The appropriate management of massive postpartum hemorrhaging remains a challenge for emergency medicine and requires a multidisciplinary team, as the prompt restoration of hemodynamic stability is important in postpartum patients. Since 1979, when pelvic arterial embolization (PAE) as a method to control postpartum hemorrhaging was first reported in literature [2], the technique has been proven to prevent surgery-associated morbidity in women who are often poor surgical candidates because of the debilitation caused by anemia and coagulation abnormalities [3] [4] [5][6]. The use of PAE with gelatin sponges in postpartum patients with hemorrhaging has increased and is a superior first-line alternative to hysterectomy for control of obstetric hemorrhaging [1]. Emergency PAE is required in patients with massive postpartum hemorrhaging because more than half of all maternal deaths occur within 24 hours of delivery, most commonly from excessive bleeding [7]. N-butyl cyanoacrylate (NBCA) almost immediately polymerizes upon contact with vascular endothelium or ionic fluids such as blood. Recently, NBCA embolization was shown to be a safe alternative treatment for the management of postpartum hemorrhaging because it works independently of the patient's and can achieve a rapid occlusion of the artery responsible for bleeding [8]. NBCA is experimentally superior to gelatin-sponges and coils with regard to hemostatic capacity [9]. In addition to the patient's skin PAE causes radiation exposure to the gonads. These are among the most radiation-sensitive organs in any individual regardless of age, sex, or fertility; the potential for malignant degeneration increases directly with the cumulative radiation dose [10]. The uterus and ovaries are in the direct path of the radiation beam throughout PAE and cannot be shielded. Despite published literatures describing radiation exposure during uterine arterial embolization for leiomyomas [7][11], no English-language studies regarding PAE in patients with postpartum hemorrhaging have been published. The purpose of this retrospective study was to evaluate radiation exposure administered during PAE used for controlling postpartum hemorrhaging in women of child-bearing age. | ||||||
|
Materials and Methods
| ||||||
|
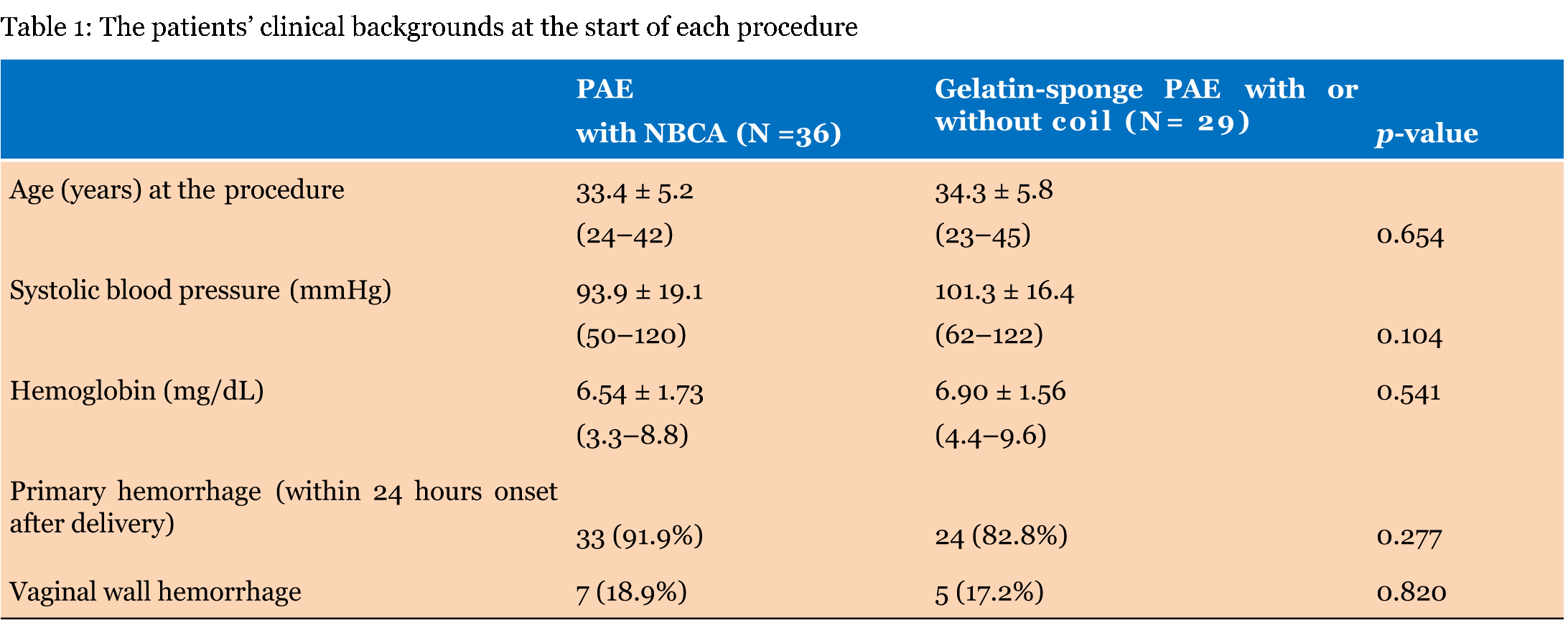
Approval for this retrospective study was obtained from our institutional review board, and a waiver for informed consent was obtained. Sixty-seven consecutive PAE procedures for controlling postpartum hemorrhaging were found by reviewing the April 2011 to May 2016 radiology database. This study consisted of 65 procedures and excluded two procedures; one was performed under a fluoroscopy in the operative room, and the other one was an abdominal wall hemorrhage after a cesarean delivery. The 65 procedures were performed in 60 female patients whose ages ranged from 23–42 years (mean 33.8 years). Hemorrhaging was atonic bleeding (n = 23) including eight post cesarean deliveries followed by incomplete uterine rupture (n = 17), vaginal tears (n = 12) including three cervical deliveries, and the retention of the placenta (n = 4). The remaining causes were placenta accreta and percreta (n = 3 in both cases) and post-hysterectomy (n = 2).Those procedures consisted of 36 PAE with and 29 PAE without NBCA. Of those procedures, two underwent intra-aortic balloon occlusion because of severe hypotension (< 50 mmHg) and a fear of cardiac arrest. PAE was performed by two board-certified radiologists who had experience with embolization in the emergency department for 18 years and 8 years, respectively. Our PAE protocol for the treatment of postpartum hemorrhaging used a right femoral arterial access with a 6-F introducer sheath (Radifocus II-H; Terumo, Tokyo, Japan) for angiography. In patients with hemodynamically unstable conditions, including < 80 mmHg of systolic blood pressure, a 5.2-F cobra occlusion catheter (Selecon MP Catheter, Terumo) with a 0.035-inch guide wire (Radifocus; Terumo co.) was used to occlude the anterior trunk of the left internal iliac arterial flow to achieve a rapid hemostasis. In patients with 70 mmHg and > 80 mmHg systolic pressure, a 4-F cobra-head catheter (Terumo Co., Tokyo Japan) was used to obtain a diagnostic internal iliac angiogram. For the selective catheterization of the uterine artery, a 2.1-F microcatheter (Sniper 2, Terumo Co.) or 2.0-F high flow microcatheter (Bobsled, Clinical supply Co., Yokohama, Japan) was passed through the parent catheter with a 0.016-inch microguidewire (Double angle-Radifocus, Terumo Co.). The right internal iliac angiography was obtained using a5-F shepherd hook catheter (Catex-CX, Gadelius Medical Co., Tokyo, Japan). The selective catheterization of the right uterine artery was performed using a coaxial catheter. For a vaginal tear, the microcatheter was selectively catheterized into the cervicovaginal, internal pudental or the obtulator artery. The gelatin sponge (Spongel; Astellas Co., Tokyo, Japan) was cut into 1- to 2- mm pieces, soaked in contrast medium (300 Img of iopamidol, Oiparomin, Konica, Tokyo Japan) and infused under fluoroscopic control. At the end of the procedure, repeat angiography was routinely performed to confirm disappearance of the intrauterine arterial branches. The adjunctive use of a coil (0.018 inch VorteX [Boston Scientific Co., Natick, MA] or a 0.035 inch Tornado [Cook C., Bloomington, Co.]) was performed to avoid recanalization of the embolized arteries when hemodynamic stability was not restored after gelatin sponge PAE. Starting in 2012, NBCA (Histoacryl B, Braun, Melsungen, Germany) was mixed with iodized oil (lipiodol: Terumo Co.) at a ratio of 1:4 (20%) and was used in postpartum patients with massive hemorrhaging. The use of those embolic materials depended on the decisions of the interventional radiologists. However, NBCA was used to exert a prompt and powerful embolic effect in patients with severe hemodynamic instability. Single gelatin sponge PAE was basically limited to mildly injured arteries without hemodynamic instability. The NBCA mixture was also used for occlusion of pseudoaneurysm and arteriovenous shunts. Following a flush of 20% glucose, 2.5 mL of 20% glue was connected to the microcatheter for the single-column injection technique. PAE procedures were performed using a single-plane digital fluoroscopic X-ray unit (Ultimax-I, Toshiba Medical Systems, Ohtawara, Japan). The control system for this equipment automatically sets the X-ray exposure kilovoltage and milliamperage. The unit used automatic selection of additional auto Tantalum filtration. For the acquisition of the images in the PAE procedures was used pulsed fluoroscopy (7.5 and 15 pulses/sec). For each PAE procedure the fluoroscopy time and the cumulative dose area-product (DAP) were registered. When the selective catheterization of either the uterine or the cervicovaginal artery failed, embolization was chosen to be distal to the origin of the posterior division of the internal iliac artery. The 65 PAE procedures were divided into two groups: (i) the procedures with NBCA (PAE with NBCA) and (ii) gelatin-sponge PAE with or without a coil(s) (PAE without NBCA). We compared the patient's clinical backgrounds before each procedure including age, systolic blood pressure, serum hemoglobin, incidences of vaginal wall hemorrhaging, and primary hemorrhaging (which was defined as blood loss > 500 mL from the genital tract within 24 h of delivery). We also compared the mean values of the number of embolized arteries as well as the mean fluoroscopy time and mean DAP between the two procedure groups. In patients who required repeated PAE, we documented hemorrhaging cause, the embolic materials used for each PAE, and cumulative DAP. Statistical analyses were performed using the Excel add-on software, Xlstat (Addinsoft, Cologne, Germany). Quantitative variables were compared with the Mann-Whitney U test, and Pearson's chi-square test was used to compare the categorical variables. In those analyses, p < 0.05 was considered statistically significant. | ||||||
|
Results | ||||||
|
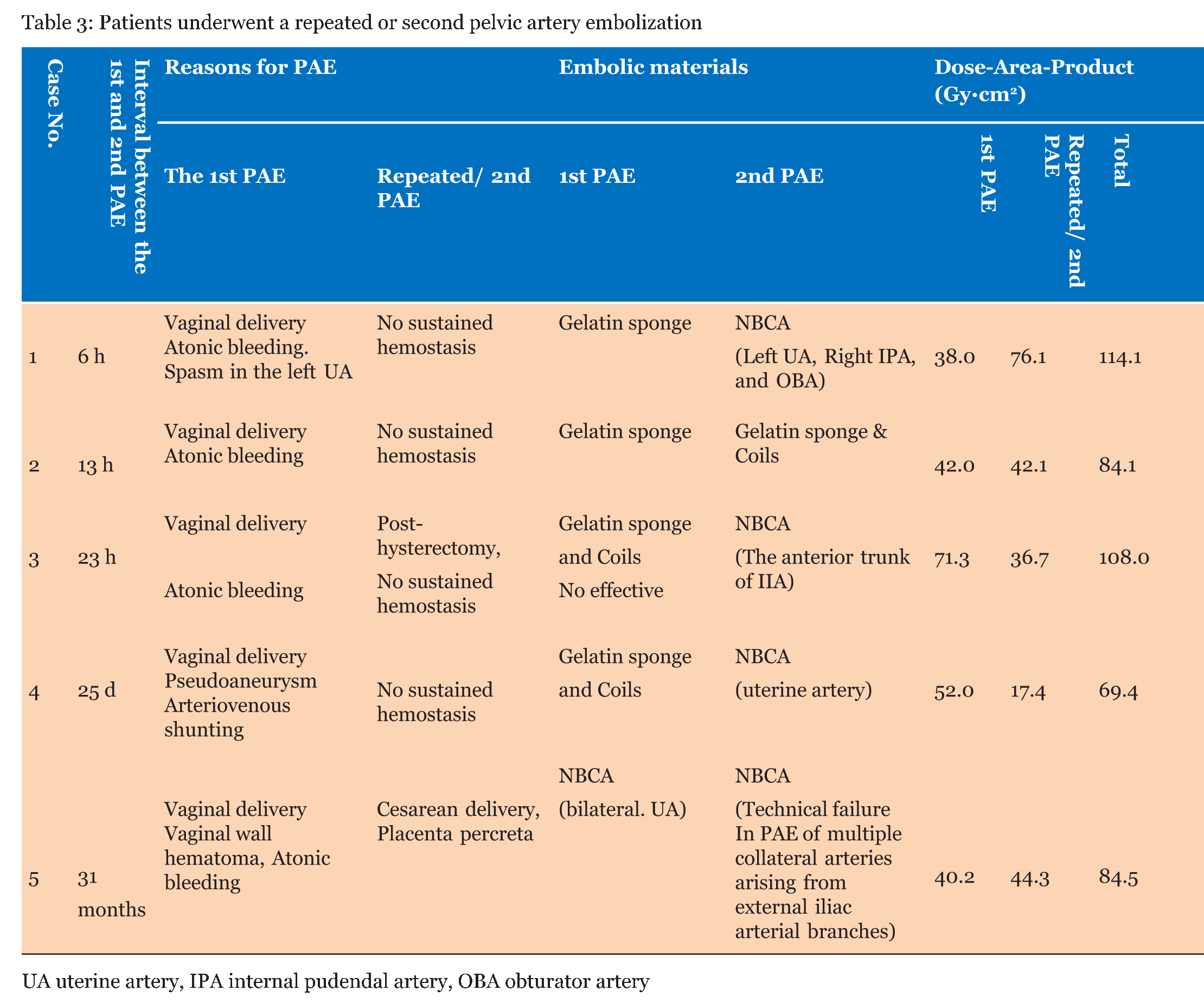
There were 36 procedures (55.4%) with and 29 (44.6%) without NBCA. Table 1 gives the patients' clinical backgrounds at the start of each procedure. No significant differences in clinical background other than the incidence of pre-embolization surgeries, which were performed in five (13.5%) procedures with NBCA and none in those without NBCA (p = 0.033) were observed. Technical success was achieved in 33 PAE with NBCA procedures (91.6%) and 26 (89.7%) of PAE with gelatin sponge and coil. Spasm of the uterine artery caused technical failures in four patients in the NBCA and three in the non-NBCA groups. Clinical success was achieved in 31 and 22 procedures (86.1% and 75.9%, respectively) in the NBCA and non-NBCA procedure groups (Table 2). There were no significant differences in success rates between the two procedures groups. In four patients, repeated PAE for recurrent hemorrhaging without concurrent restoration of hemodynamic instability was performed after the PAE without NBCA procedure; repeated procedures were NBCA procedures in three patients and gelatin sponge in combination with a coil in one patient (Table 3). Thirty-one months after the initial PAE that occluded the bilateral uterine arteries with NBCA, the remaining patient underwent a second PAE due to cesarean delivery and placenta percreta, a hysterectomy was performed after technical failure of the second embolization of multiple tiny collateral ways from the medial collateral arteries of the deep femoral arteries. Cumulative DAP was 69.4–114.1 Gy·cm2 (mean, 92.0±18.5 Gy·cm2) in five cases who underwent the second PAE, including four patients requiring repeated procedures within 6 h – 25 days after the initial PAE. | ||||||
| ||||||
| ||||||
|
| ||||||
| ||||||
|
Discussion
| ||||||
|
Although PAE for controlling postpartum hemorrhaging has been widely accepted as the first-line treatment, there have been some studies that have assessed radiation exposure to the patient that occurs during the procedure [12]. For interventional radiologists, long-term effects of radiation exposure to the woman are secondary considerations because PAE for postpartum hemorrhaging, unlike PAE for the uterine fibroid, is of utmost importance in order to save a woman's life. However, they should be concerned about whether the radiation exposure may cause any damage to the child-bearing woman because it appears that in the hands of the experienced interventional radiologists, short-term outcomes of embolization in terms of saving life are quite good. It is of particular interest that increased doses to the ovaries in women of childbearing age who underwent PAE may causes genetic injury and increase the risk of radiation-induced cancer in later life. Interventional radiologists, as well as their radiological technologists should be aware of the potential for injury from these procedures. For purposes of optimization of the practices on this procedures, several preventative guidelines should be followed:
The use of pulsed versus nonpulsed fluoroscopy during interventional procedures was shown to reduce radiation exposure. Andrews et al. reported that the use of gelatin sponge PAE for the treatment of uterine fibroids required 14.2 min for the mean fluoroscopic times and 30.5 Gy·cm2 for the mean DAP [11]. This study of gelatin sponge PAE with or without a coil(s) for controlling postpartum hemorrhaging required longer mean fluoroscopic times (34.2 minutes) and greater mean DAP (55.1 Gy·cm2). The reason for the longest fluoroscopy time in this study is due to an infusion of gelatin sponge under fluoroscopic guidance that required a longer fluoroscopic time through the coaxial catheter than through a 4- or 5-F catheter. Our PAE protocol for controlling postpartum hemorrhaging included a selective catheterization with a coaxial catheter for uterine or other responsible arteries, because catheterization with a 4- or 5-F catheter into the uterine artery often caused arterial spasms in hemorrhagic patients. This PAE with NBCA study required shorter mean fluoroscopic times (26.3 minutes) and smaller mean DAP (43.5 Gy·cm2) than PAE with gelatin-sponges. A complete occlusion of the uterine artery or other branches of the internal iliac artery was achieved by a single infusion which required approximately 10 sec of fluoroscopic time, although an infusion of NBCA-lipiodol mixture was also performed through a coaxial catheter under a fluoroscopic guidance. Nikolic et al. placed the thermoluminescent dosimeter into the posterior fornix of the vagina and on the skin at the beam entrance site for the uterine fibromas; they reported that the mean estimated absorbed each skin and ovarian dose was measured as 1.66 Gy and 0.22 Gy, respectively with 21.9 minutes of the mean fluoroscopic time [7]. The mean value of the ovarian dose was approximately 10 times of the assessed absorbed ovarian dose during computed tomography of the trunk (0.02 Gy). Fallopian tube recanalization (0.2–2.75 Gy), and computed tomography of the trunk (0.1–1.9 Gy). PAE using NBCA as the primary embolic agent is an effective method for treating primary postpartum hemorrhage with extravasation or pseudoaneurysm [8] [13]. The NBCA is a permanent embolic material and is expected to be superior to gelatin sponge and coils because it works independently of the patient's hemostatic capacity. Hemostatic durability is more effective than gelatin sponge. Yonemitsu et al. reached arterial branches < 300 mm in diameter [9]. The mechanism of hemostasis by gelatin sponge or coils involves thrombus formation, which depends on the patient's hemostatic capacity for successful embolization. However, severe coagulopathy, which is frequently associated with life-threatening massive hemorrhaging in postpartum patients, might prohibit thrombus formation and, thus, the achievement of hemostasis might fail. Although a hysterectomy does not always result in hemostasis and precludes future pregnancy, PAE may achieve hemostasis with preservation of the patient's reproductive capacity. Although PAE's long-term effects on a woman's ability to have a child are unknown, pregnancies of women who were treated with gelatin-sponge uterine embolization for fibroids were at significantly increased risk for spontaneous abortion (22%), preterm delivery (22.2%), a 17% rate of mal-presentation, and a 7% rate of small for gestational age [14]. Holub et al. also reported that pregnancies after uterine embolization with gelatin sponge had a statistically significantly higher rate for spontaneous abortion (56%) than those pregnancies after surgical uterine artery occlusion (10.5%) [15]. In women who undergo gelatin sponge PAE, ischemia and adverse effect of gelatin sponges on their uterine tissues may cause spontaneous abortion. In this study, there was a woman who was treated with NBCA embolization had a pregnancy with placenta percreta, and underwent massive hemorrhaging after cesarean delivery. Instead of using gelatin sponge, coil, or NBCA PAE secondary embolization for massive hemorrhaging after the next delivery, we recommend hysterectomy rather than the second PAE in those cases. This study does have a number of limitations. First, the number of patients in this study was small. Second, PAE with gelatin sponge alone could not be evaluated without separating gelatin-sponge in combination with coil. Third, we did not measured estimated skin or ovarian absorbed dose during PEA. However, this study showed PAE combined with NBCA was superior in reduction of radiation exposure compared to the procedure with gelatin sponge of a coil(s) in patients with postpartum hemorrhage. Interventional radiologists should take long-term effects of radiation exposure to woman who underwent PAE for controlling postpartum hemorrhaging. Radiologists should avoid under-evaluating radiation exposure during treatment. Patients counseling should be undertaken routinely and with appropriate follow-ups as recommended by the International Commission on Radiological Protection [10] , especially in patients who have undergone a second or repeated PAE. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions:
Izumi Torimoto – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Shigeo Takebayashi – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Zenjiro Sekikawa – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Akitoshi Nishii – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Soichiro Obata – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Shigeru Aoki – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2016 Izumi Torimoto et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|